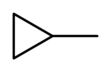
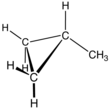
Methylcyclopropane
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Methylcyclopropane | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.008.934 | ||
| EC Number |
| ||
| MeSH | C105498 | ||
PubChem CID |
| ||
| UNII |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | C4H8 | ||
| Molar mass | 56.108 g·mol−1 | ||
| Appearance | Colourless gas[1] | ||
| Density | 0.6912 g/cm3[1] | ||
| Melting point | −177.3 °C (−287.1 °F; 95.8 K)[1] | ||
| Boiling point | 0.7 °C (33.3 °F; 273.8 K)[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |||
Chemical compound
Methylcyclopropane is an organic compound with the structural formula C3H5CH3. This colorless gas is the monomethyl derivative of cyclopropane.
Reactions
Methylcyclopropane, like many other cyclopropanes, undergoes ring-opening reactions. Bond cleavage in certain reactions is also reported in conjunction with the use of methylenecyclopropane groups as protective groups for amines.[citation needed]
References
- ^ a b c d Lide, David. R, ed. (2009). CRC Handbook of Chemistry and Physics (89th ed.). CRC Press. ISBN 978-1-4200-6679-1.
- v
- t
- e
aliphatic
hydrocarbons
| Alkanes CnH2n + 2 |
| ||||
|---|---|---|---|---|---|
| Cycloalkanes | |||||
| Alkylcycloalkanes |
| ||||
| Bicycloalkanes |
| ||||
| Polycycloalkanes |
| ||||
| Other |
|
aliphatic
hydrocarbons
| Alkenes CnH2n |
| ||||
|---|---|---|---|---|---|
| Alkynes CnH2n − 2 |
| ||||
| Cycloalkenes | |||||
| Alkylcycloalkenes |
| ||||
| Bicycloalkenes | |||||
| Cycloalkynes |
| ||||
| Dienes |
| ||||
| Other |
|
hydrocarbons
| PAHs |
| ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alkylbenzenes |
| ||||||||||||||||||||||
| Vinylbenzenes | |||||||||||||||||||||||
| Other |
|